2017 - IPITA
This page contains exclusive content for the member of the following sections: TTS, IPITA. Log in to view.
Alternative Islet Sources
7.3 - Ischemia response of human stem cell-derived insulin-producing cells
Presenter: GF, Faleo, San Francisco, United States
Authors: Gaetano Faleo, Steven Wisel, Audrey Parent, Holger Russ, Matthias Hebrok, Qizhi Tang
Ischemia response of human stem cell-derived insulin-producing cells
G. Faleo1, S. Wisel1, A. Parent2, H. Russ2, M. Hebrok2, Q. Tang1,2.
1Department of Surgery, UCSF, San Francisco, USA, ; 2Diabetes Center, UCSF, San Francisco, USA,
Background: Ischemia dramatically impairs function and survival of transplanted islets. Additionally, ischemic islets produce mediators that contribute to revascularization, but also contribute to leukocyte recruitment and inflammation. Human Stem Cell-derived Insulin-Producing Cells (SCIPC) are poised to replace donor-derived islets as a source of beta cells for transplantation. However, the effects of ischemia on SCIPCs after transplantation have not been well characterized. The objective of this study is to better understand SCIPC response to ischemia to inform future development of strategies to optimize SCIPC survival, differentiation, and function after transplant.
Methods: We generatedSCIPC from stem cells that have a GFP transgene in the insulin locus and a constitutively expressed luciferase transgene so that more mature insulin-producing cells and total viable cell mass can be tracked using GFP and luciferase. SCIPC were transplanted under the kidney capsule of NSG mice and monitored graft mass using luciferase bioluminescence. Some grafts were recovered on different days after transplant and analyzed using flow cytometry, immunofluorescence microscopy, and RNA expressing by nanostring. Individual contribution of hypoxia and nutrient deprivation on SCIPC were investigated in vitro with exposure to 1% oxygen, nutrient starvation or both for 24 hours. Cells were examined for viability, RNA expression, cytokine production using luminex, and glucose-stimulated insulin secretion (GSIS) to assess function.
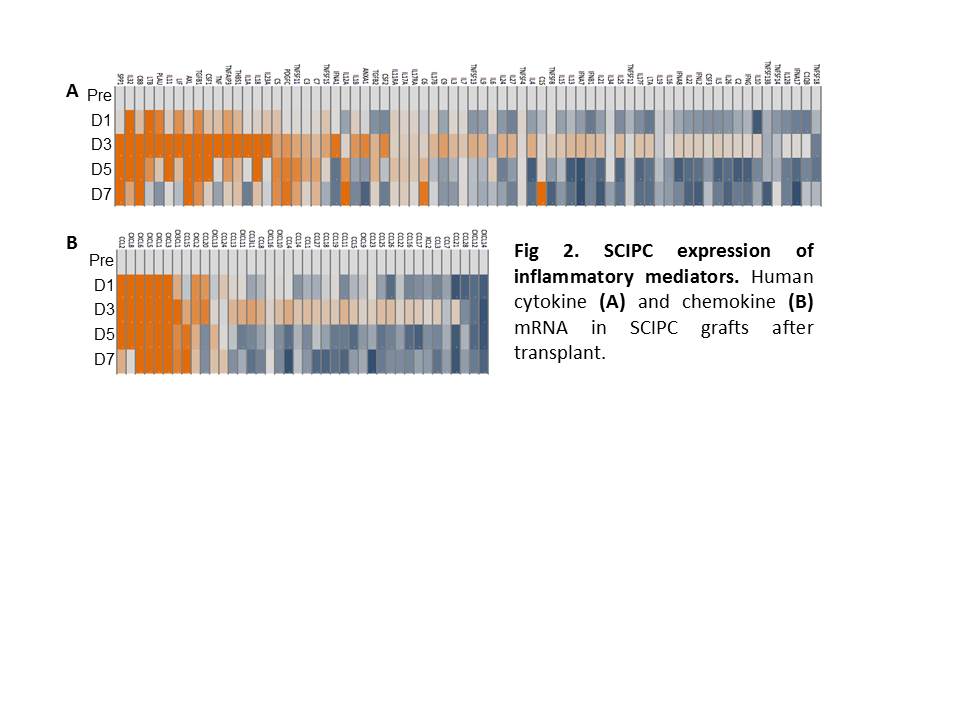
Results: SCIPC mass decreased by 40% seven days after transplant. GFP+ fraction dropped from 38% pre-transplant to 12% and pancreatic progenitor marker Pdx1 decreased from 95.4% to 77.7% suggesting preferential death of mature cells (Fig.1A and B). In vitro analyses show that SCIPC survival was not greatly affected by hypoxia or starvation alone but presence of both stressors synergistically killed SCIPC (Fig.1C). Cells survived hypoxia showed reduction of INS, GCG, and SST gene expression and upregulation of HIF-target genes GLUT1, HK1, HK2, PDK1, LDHA, MCT4, and IRS1. GSIS index declined from 1.8 to 1.2 after hypoxia exposure (Fig.1D). mRNA analysis of SCIPIC grafts using Nanostring Cancer Immunology 800 gene panel revealed induction of human cytokines and chemokines including IL1, SPP1, CCL2, CXCL8 after transplant (Fig 2), which was associated with macrophage infiltration as early as day 3 after transplant. SCIPC production of inflammatory mediators was corroborated by the finding that SCIPCs cultured in vitro secreted CCL2, CXCL8, CXCL12, and CXCL1.
Conclusions: Our results show that ischemia can kill SCIPC and those survive have a less mature phenotype. Hypoxia exposure in vitro led to a change in expression of glucose transporters and switch to glycolytic metabolism by SCIPC and loss of GSIS response. SCIPIC also produce inflammatory mediators in response to ischemia, which may contribute to leukocyte recruitment and graft failure after transplant. Strategies that minimize ischemia response by SCIPC will be important for realizing the therapeutic potential of stem cell based therapy for diabetes.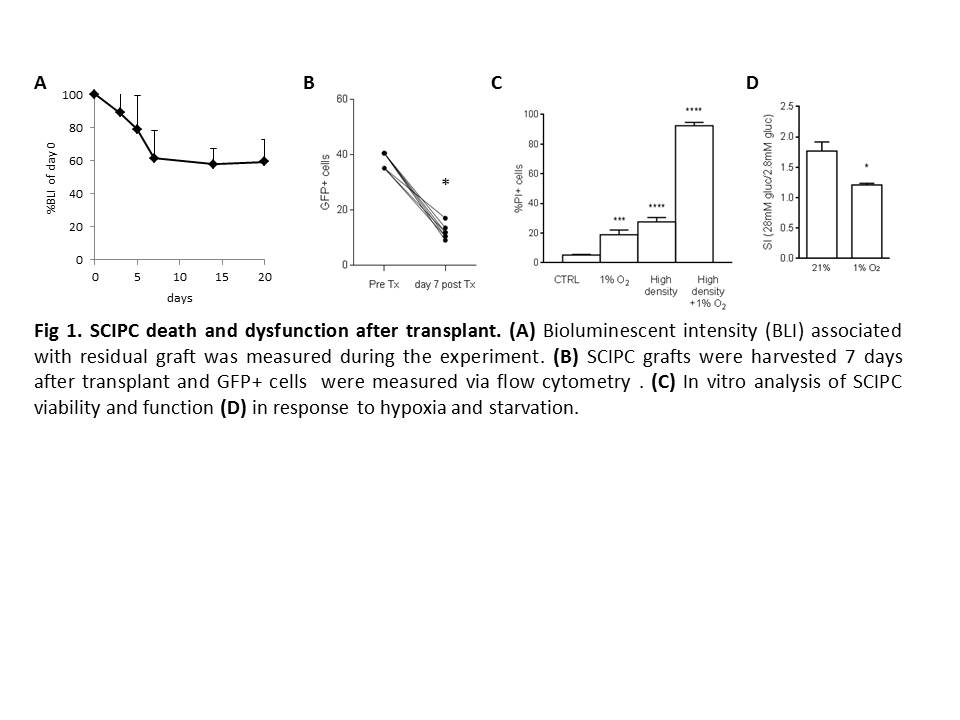
Important Disclaimer
By viewing the material on this site you understand and accept that:
- The opinions and statements expressed on this site reflect the views of the author or authors and do not necessarily reflect those of The Transplantation Society and/or its Sections.
- The hosting of material on The Transplantation Society site does not signify endorsement of this material by The Transplantation Society and/or its Sections.
- The material is solely for educational purposes for qualified health care professionals.
- The Transplantation Society and/or its Sections are not liable for any decision made or action taken based on the information contained in the material on this site.
- The information cannot be used as a substitute for professional care.
- The information does not represent a standard of care.
- No physician-patient relationship is being established.
Contact
Address
The Transplantation Society
International Headquarters
740 Notre-Dame Ouest
Suite 1245
Montréal, QC, H3C 3X6
Canada
