DECEASED ORGAN DONATION AND RELIGION SYMPOSIUM - MARCH 12-14, 2020 - ANKARA, TURKEY
Hotel rates start at $35 USD per night for single accommodation at the Symposium Hotel.
IPTA OUTREACH PROGRAM 2020 - APPLICATIONS NOW OPEN!
Dear Colleagues,
The IPTA Outreach Committee is pleased to announce that applications for the 2020 Outreach Program are open. The program supports emerging centres that seek to develop or expand/enhance care in existing transplant programs.
Usually, an emerging centre will have identified a suitable supporting centre to assist them with this program; however it is possible the Outreach Committee can assist in this process.
The application package, guidelines and useful reference documents are available on the Outreach Program section of the IPTA website.
CLICK HERE TO VISIT THE WEBSITE
With many thanks,
Fiona Mackie
Chair, IPTA Outreach Committee
TTS-ILTS PAIRED TRANSPLANT CENTERS PROGRAM
The TTS-ILTS Paired Transplant Centers Program is a collaboration between The Transplantation Society (TTS) and the International Liver Transplantation Society (ILTS) supporting new liver transplant programs in emerging countries.
YOUNG MEMBERS' SCIENTIFIC AWARDS

In order to help young members offset expenses to attend our Congress in Seoul, TTS 2020 will be offering Young Investigator Awards and International Transplantation Science Mentee-Mentor Awards. Award recipients will be selected on scientific merits (abstract submission deadline February 18, 2020).
Young Investigator Awards
To be eligible for the Young Investigator Awards, you need to apply by March 23, 2020, submit an abstract to the Congress, be a trainee (within 2 years of completion of their training and/or fellowship) and be a member of TTS.
International Transplantation Science Mentee-Mentor Awards
TTS in collaboration with National and International Societies acknowledges the contribution of basic science to the field of transplantation by offering Mentee-Mentor awards.
To be eligible, you need to apply by March 23, 2020, submit an abstract to the Congress, be a graduate student or trainee (within 5 years of receiving PhD, MD), both Mentee and Mentor must attend TTS 2020 and be member of both TTS and confirmed supporting Societies.

TRANSPLANTATION - HIGHLIGHTED ARTICLE

Dr. Karen Keung, Editorial Fellow, Transplantation
Conversion From Calcineurin Inhibitors to Belatacept in HLA-sensitized Kidney Transplant Recipients With Low-level Donor-specific Antibodies
Ullo CA, Anglicheau D, Snanoudj R, et al.
Transplantation: October 2019 - Volume 103 - Issue 10 - p 2150–2156
Data on the conversion to Belatacept of HLA-sensitised patients with DSAs in renal transplantation is limited. In this article, the authors present their centre's experience with 29 DSA-positive transplant recipients who were switched from a CNI-based regimen to Belatacept and report on acute rejection incidence, DSAs and renal function.They were compared to a cohort of 42 non HLA-sensitised (DSA-negative) patients switched to belatacept during the same period; conversion in both groups was based on suspected CNI toxicity on biopsy.
Conversion to belatacept occurred at a median of 444 days (IQR, 85-1200) after transplantation and patients were followed up for a median of 308 days (IQR, 125-511). At the time of conversion, 16 of the 29 DSA-positive patients had a circulating DSA. None in this group developed de novo DSAs, and at the end of follow up pre-existing DSAs had decreased or stabilized. In the DSA negative group, 1 patient developed a de novo class I DSA. No significant difference between groups in terms of eGFR, proteinuria, or biopsy-proven acute rejection (1 and 2 cases in the DSA-positive and DSA-negative groups respectively) were observed.
This data supports the safety of Belatacept conversion in transplant patients with low-MFI DSA, although further prospective evaluation in larger cohorts is warranted.
TTS2020 - ABSTRACT SUBMISSION OPEN!
If you are a TTS Member, we have setup an account for you in the system. You can retrieve your login credentials in the TTS Members area of tts.org.
OPTN FEEDBACK REQUEST
The OPTN is seeking feedback on initiatives impacting organ donation and transplantation in the U.S. The public comment period for the initiatives below will be open from January 22, 2020 to March 24, 2020. You can review proposals and submit your approval, disapproval, support, lack of support or suggestions on the OPTN Public Comment Website.
TTS CALL FOR NOMINATIONS (PRESIDENT-ELECT, SECRETARY, TREASURER AND COUNCIL)

In 2020, three Officer positions will be vacated and 7 of the 12 Councilors-at-large representing the Regions will be changing. The elections will take place early in 2020 and those elected will assume their new roles starting at the 2020 TTS Congress in Seoul.
Members can access the online nominating form at www.tts.org/nominations. Since each nominee must have his or her form signed by three supporting members (including him/herself), the online process allows for efficient and rapid circulation.
THE NOMINATION DEADLINE IS FEBRUARY 14, 2020.
Please note:
- As successive presidents may not be from the same Region, members from the North America region who would have otherwise been eligible to become President-Elect are not eligible in these elections.
- Only full members who have served a full term on Council and have paid their dues are eligible for the Officer positions (President-Elect, Secretary and Treasurer).
- Only full members who have paid these dues are eligible to be nominated for Council.
- Only full members who have paid their dues may nominate and/or vote.
For more information on elections, visit the TTS website and consult the By-Laws in the “About” section.
Nominations are being sought for these positions
Officer Positions:
- President-Elect
- Secretary
- Treasurer
TTS Regions:
- Asia (2)
- Europe (1)
- Latin America (2)
- North America (2)
WEDNESDAY, FEBRUARY 12 - WEBINAR ANNOUNCEMENT
TITLE: DONOR CELL FREE DNA TESTING
WEDNESDAY, FEBRUARY 12, 2020 - 1:00 PM EST (MONTREAL TIME)
Objectives:
- Explain how donor cell free DNA testing is done and the different methods.
- Examine how it is different in different organs and in plasma vs. urine.
TTS Education Committee's Precision Medicine Webinar Series - NEW RELEASE
New release - the most recent recording in the Precision Medicine Webinar Series. Dr. Bruce Kaplan and Dr. Mark Stegall discuss principals of evidence based medicine as it applies to biomarkers and clinical trial design. Still available, the previous webinars in the series. Please note this recording is without slides and is discussion based.
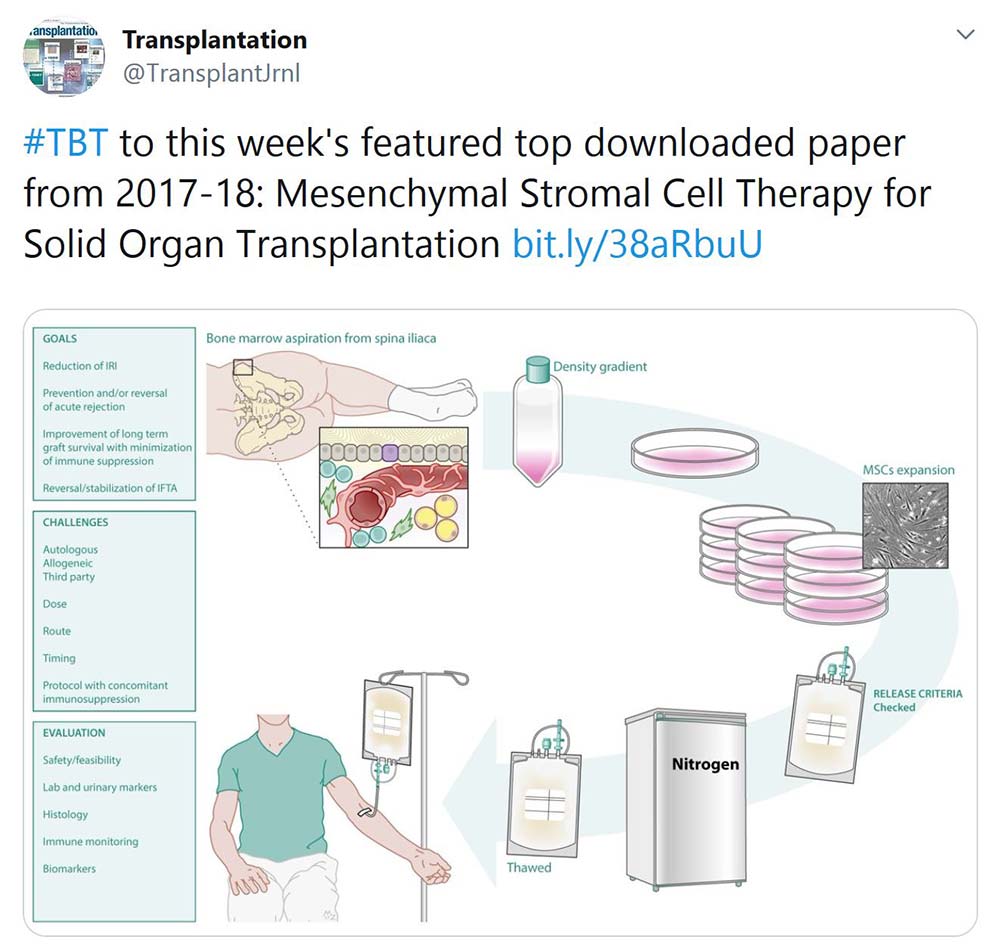
TRANSPLANTATION - WEEK'S MOST DOWNLOADED PAPER
IN THE NEWS
JAPAN RESEARCHERS CONDUCT WORLD'S 1ST TRANSPLANT OF IPS HEART MUSCLES
Jan. 27 - A research team led by a Japanese medical school professor said Monday it has conducted the world's first transplant of heart muscle tissues using artificially derived stem cells known as iPS. Yoshiki Sawa of Osaka University's medical school said the transplant was conducted Monday in a clinical trial to study the treatment's effectiveness and safety in a patient with serious heart failure.
INITIAL BIOPSY OF DECEASED DONOR KIDNEYS NOT RELIABLE FOR PREDICTING POST-TRANSPLANT OUTCOMES
Jan. 23 - Initial biopsies of deceased donor kidneys, also known as procurement biopsies, often provided information that was neither reproducible nor associated with allograft outcomes, according to a recently published study. Therefore, researchers contended that procurement biopsy results should not be the deciding factor as to whether a kidney is suitable for transplant.
TINY ORGANS GROWN FROM SNAKE GLANDS PRODUCE REAL VENOM
Jan. 23 - Venomous snakes kill or permanently injure more than a half-million people every year. Yet researchers still know surprisingly little about the biology behind venom, complicating efforts to develop treatments. A new advance could help: Researchers have successfully grown miniature organs from snake stem cells in the lab that function just like snake venom glands; they even produce real venom.
TRANSPLANT IN MULTIPLE MYELOMA DEEMED SAFE IN OLDER PATIENTS — BUT IS IT NECESSARY?

Jan. 27 - Despite the rapidly changing landscape of treatments available for multiple myeloma, autologous stem cell transplant (ASCT) remains a standard of care among eligible patients, with median overall survival as long as 10 years among standard-risk patients.
UPCOMING MEETINGS AND ANNOUNCEMENTS
4th International Chauvet Workshop - Save the Date!
We are excited to announce that the planning for the Chauvet 2020 meeting is well underway. This meeting is of interest to all health care providers who care for individuals with upper extremity amputations, severe facial disfigurement, absolute uterine factor infertility and those with loss of laryngeal function and all VCA candidates and recipients. If you are in any discipline with an interest in the psychosocial aspects of these patients, this meeting will interest you.
MEETING FACTS
- Location: Mayo Clinic Downtown Campus, Rochester MN, USA
- Date: April 30 (beginning at 12pm CDT) to May 2 (ending at 5pm).
- Registration fee: $310 (Includes meeting registration, two breakfasts and lunches and two gala dinners and networking events at the historic homes of Drs. William and Charles Mayo,)
- Lodging: There are several reasonably priced hotels in close proximity to the meeting location.
Email us to add yourself to our mailing list or with questions at transplant-rst@mayo.edu.
Watch for links to more information coming soon!
3rd IPITA/JDRF/HSCI Conference on Stem Cell Derived Beta Cells
18th Asian Pacific Congress of Nephrology (APCN)
Contact
Address
The Transplantation Society
International Headquarters
740 Notre-Dame Ouest
Suite 1245
Montréal, QC, H3C 3X6
Canada
Используйте Вавада казино для игры с бонусом — активируйте промокод и начните выигрывать уже сегодня!