
TTS 2020 Virtual Congress - A Virtual Learning Experience

JUST RELEASED - TRANSPLANTATION - SEPTEMBER ISSUE
This month is focused almost entirely on the issue of ischaemia/reperfusion and the science and mechanisms of preservation. Some great reviews and commentaries place this area of research into context describing the routes to new applications, new therapeutics, and better use of the knowledge we do have, as well as pointing to the knowledge we need. While there has been much focus on the clinical progress in applications of the various options for liver and kidney normothermic and hypothermic preservation, there are also much needed considerations of the issues in intestinal, lung, and heart preservation. Finally, a reminder of the sensitivity of neuronal tissues to ischaemia after pancreas transplantation. A month of reading that you cannot miss.
CLICK HERE TO ACCESS THIS ISSUE
TTS MEMBERS - CLICK HERE TO SIGN-IN FOR OPEN ACCESS THROUGH TTS.ORG
TRANSPLANTATION DIRECT - HIGHLIGHTED ARTICLE
Dr. Jeremy R. Chapman, Editor-in-Chief, Transplantation
Donor-derived Cell-free DNA Combined With Histology Improves Prediction of Estimated Glomerular Filtration Rate Over Time in Kidney Transplant Recipients Compared With Histology Alone
Huang E, Gillespie M, Ammerman N, et al.
Transplantation Direct: August 2020 - Volume 6 - Issue 8 - p e580
It is now very clear that kidney transplant biopsies with more ‘Banff’ classified inflammation and with higher chronicity scores do worse. These investigators asked the question: is it possible to improve prediction of renal outcomes by adding data from tests of donor-derived cell-free DNA? In 180 kidney transplant patients with dd-cfDNA assessed within 1 month of a biopsy they used multiple statistical methods to assess whether prediction is improved by use of both measures. A model including dd-cfDNA with the Banff scores cg, ci + ct with covariate-by-time interactions had a better fit compared with cg, ci + ct alone in predicting eGFR. It is tempting to extrapolate these findings to suggest that chronic inflammation that both destroys donor cells and invokes a fibrotic response does the most damage. This hypothesis needs testing in other centres and using other techniques but inches us forward if true.
JUST RELEASED - TRANSPLANTATION DIRECT - SEPTEMBER ISSUE
A full spectrum of topics appears in the September issue of Transplantation Direct. The issue starts with an interesting case series on the use of patiromer to treat hyperkalemia in transplant recipients. Regarding normothermic organ perfusion research (kidney, liver and lung), there are articles on using flavin mononucleotide as a marker of organ damage, and on the feasibility of ex vivo arterial reconstruction surgery for liver grafts. In liver transplantation, there are reports on using non-interruptive intraoperative transesophageal echocardiography to assess for hepatic vascular structures, and on the impact of the 2015 T2 hepatocellular carcinoma exception policy on geographic variability and explant tumor viability assessments. In pediatric liver transplantation, the use of low versus high dose rabbit ATG is assessed for induction therapy. Related to islet and pancreas transplantation, the effectiveness of trametinib (MEK inhibitor) on murine islet rejection is assessed (with mechanistic studies), and SRTR data is analyzed for the impact of functional status on survival in simultaneous pancreas-kidney transplant recipients. For those interested in vascularized composite tissue transplantation, a new rat model is presented for hind-limb transplantation that importantly includes the draining lymphatic tissues to assess the role of the lymphatic system. In the area of organ donation, the Live Donor Champion Program in kidney transplantation looks at the effectiveness of the program in promoting live donation in populations with historically lower access to organ transplantation. For more details about this issue, please go to our open access Transplantation Direct website.
TRANSPLANTATION - WEEK'S MOST DOWNLOADED PAPER
«HOT OFF THE PRESS»
RECENT PUBLICATIONS IDENTIFIED BY TTS EDUCATION COMMITTEE ON COVID-19
Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis
The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group.
JAMA. Published online September 02, 2020. doi:10.1001/jama.2020.17023
The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT)Working Group This is a prospective, meta-analysis that pooled data from 7 randomized clinical trials that evaluated the efficacy of corticosteroids in 1703 critically ill patients with COVID-19. The trials were conducted in 12 countries from February 26, 2020, to June 9, 2020. Patients had been randomized to receive systemic dexamethasone, hydrocortisone, or methylprednisolone (678 patients) or to receive usual care or placebo(1025 patients). There were 222 deaths among the 678 patients randomized to corticosteroids and 425 deaths among the 1025 patients randomized to usual care or placebo (summary OR, 0.66 [95%CI, 0.53-0.82]; P < .001 based on a fixed-effect meta-analysis). The fixed-effect summary OR for the association with mortality was 0.64 (95%CI, 0.50-0.82; P < .001) for dexamethasone compared with usual care or placebo (3 trials, 1282 patients, and 527 deaths), the OR was 0.69 (95%CI, 0.43-1.12; P = .13) for hydrocortisone (3 trials, 374 patients, and 94 deaths), and the OR was 0.91 (95%CI, 0.29-2.87; P = .87) for methylprednisolone (1 trial, 47 patients, and 26 deaths). In this prospective meta-analysis of clinical trials of critically ill patients with COVID-19, administration of systemic corticosteroids, compared with usual care or placebo, was associated with lower 28-day all-cause mortality.
Effect of Dexamethasone on Days Alive and Ventilator-Free in Patients With Moderate or Severe Acute Respiratory Distress Syndrome and COVID-19 The CoDEX Randomized Clinical Trial
Tomazini BM, Maia IS, Cavalcanti AB, et al.
JAMA. Published online September 02, 2020. doi:10.1001/jama.2020.17021
This is a multicenter, randomized, open-label, clinical trial conducted in 41 intensive care units (ICUs) in Brazil. Twenty mg of dexamethasone intravenously daily for 5 days, 10mg of dexamethasone daily for 5 days or until ICU discharge, plus standard care (n =151) or standard care alone (n = 148). Patients randomized to the dexamethasone group had a mean 6.6 ventilator-free days (95%CI, 5.0-8.2) during the first 28 days vs 4.0 ventilator-free days (95%CI, 2.9-5.4) in the standard care group (difference, 2.26; 95%CI, 0.2-4.38; P = .04). There was no significant difference in the prespecified secondary outcomes of all-cause mortality at 28 days, ICU-free days during the first 28 days, mechanical ventilation duration at 28 days, or the 6-point ordinal scale at 15 days. Thirty-three patients (21.9%) in the dexamethasone group vs 43 (29.1%) in the standard care group experienced secondary infections, 47 (31.1%) vs 42 (28.3%) needed insulin for glucose control, and 5 (3.3%) vs 9 (6.1%) experienced other serious adverse events.
Humoral Immune Response to SARS-CoV-2 in Iceland
D. F. Gudbjartsson et al.
N. Engl. J. Med., p. NEJMoa2026116, Sep. 2020, doi: 10.1056/NEJMoa2026116.
This study measured antibodies in serum samples from 30,576 persons in Iceland, using six assays (including two pan-immunoglobulin [pan-Ig] assays). The authors tested 2102 samples collected from 1237 persons up to 4 months after diagnosis by a quantitative polymerase-chain-reaction (qPCR) assay. The authors measured antibodies in 4222 quarantined persons who had been exposed to SARS-CoV-2 and in 23,452 persons not known to have been exposed. Of the 1797 persons who had recovered from SARS-CoV-2 infection, 1107 of the 1215 who were tested (91.1%) were seropositive, antiviral antibody titers increased during 2 months after diagnosis by qPCR and remained on a plateau for the remainder of the study. Of quarantined persons, 2.3% were seropositive; of those with unknown exposure, 0.3% were positive. The authors estimated that 0.9% of Icelanders were infected with SARS-CoV-2 and that the infection was fatal in 0.3%. 56% of all SARS-CoV-2 infections in Iceland had been diagnosed with qPCR, 14% had occurred in quarantined persons who had not been tested with qPCR (or who had not received a positive result, if tested), and 30% had occurred in persons outside quarantine and not tested with qPCR.
Sex differences in immune responses that underlie COVID-19 disease outcomes
Takahashi, T., Ellingson, M.K., Wong, P. et al.
Nature (2020). https://doi.org/10.1038/s41586-020-2700-3
This study examined sex diferences in viral loads, SARS-CoV-2-specifc antibody titers, plasma cytokines, as well as blood cell phenotyping in COVID-19 patients. By focusing on patients with moderate disease who had not received immunomodulatory medications, the results revealed that male patients had higher plasma levels of innate immune cytokines such as IL-8 and IL-18 along with more robust induction of non-classical monocytes. In contrast, female patients mounted significantly more robust T cell activation than male patients during SARS-CoV-2 infection, which was sustained in old age. Importantly, a poor T cell response negatively correlated with patients’ age and was associated with worse disease outcome in male patients, but not in female patients. Conversely, higher innate immune cytokines in female patients associated with worse disease progression, but not in male patients.
CORONAVIRUS (COVID-19) UPDATES
INVITATION TO PARTICIPATE

Dear Members,
Researchers at McGill University are currently conducting a study entitled LIST-COVID-19 to study the impact of the COVID-19 pandemic on transplantation activity, volumes and immunosuppression practices on a global scale. The study will also help better understand the current and anticipated risks to transplantation during the pandemic and inform clinical practice during the “ramp-up” phase. The Transplantation Society is supporting the project, as well as several National and International Societies.
They are looking for physicians who take care of patients with a solid organ transplant to take a 10-15 minute survey on behalf of their center and program. Please note this is not a patient registry and no specific patient-level data is being collected. Please be assured they will only perform a collective analysis of the survey questions and no personal information identifying the participants will be divulged. The survey responses will be confidential. If you are interested in participating, please contact them at the emails listed below.
Investigators: Shaifali Sandal, MD and Marcelo Cantarovich, MD
Contact: shaifali.sandal@mcgill.ca and marcelo.cantarovich@muhc.mcgill.ca
The Transplantation Society (TTS) and our journal Transplantation have developed online resources to keep you informed on the Coronavirus (COVID-19) outbreak.
- TTS Coronavirus (COVID-19) Dashboard
www.tts.org/covid-19 - Transplantation Global Transplantation COVID Report
www.tts.org/txjcovid19
We are also requesting contributions and news from the transplant community to be sent to covid-19@tts.org for inclusion on our resources page.
In this dashboard, you will find links to TTS and other global and regional resources, as well as interactive maps, publications and webinars. We encourage you to explore this dashboard and share with your colleagues.
Editors and contributors to Transplantation have shared their thoughts on how they are dealing with the current crisis. While we understand that the information of today may be quite different tomorrow in this fast-moving pandemic, this report will open our forum of an international exchange on COVID for the transplant community.
Website - www.tts.org/txjcovid19
Please send your own contributions and news to covid-19@tts.org for inclusion on our resources page.
IN THE NEWS
BIG STEP IN REGENERATIVE MEDICINE
Sept. 1 - Harvard Medical School surgeons at Massachusetts Eye and Ear have replaced the ocular surface of four patients who each experienced chemical burns to one eye. The technique, cultivated autologous limbal epithelial cell transplantation (CALEC), uses stem cells taken from a patient’s healthy eye. These four cases are part of an ongoing clinical trial supported by the National Eye Institute and represent the first procedures of their kind to occur in the United States.
HEPATITIS C-INFECTED KIDNEYS CAN BE SAFELY TRANSPLANTED INTO HEALTHY RECIPIENTS
Sept. 2 - Kidneys from deceased donors with hepatitis C virus (HCV) infection can be safely transplanted into non-infected recipients when a regimen of direct-acting antiviral therapies is initiated as early as two days after the transplant, according to a clinical trial study “Multicenter Study to Transplant Hepatitis C–Infected Kidneys (MYTHIC): An Open-Label Study of Combined Glecaprevir and Pibrentasvir to Treat Recipients of Transplanted Kidneys from Deceased Donors with Hepatitis C Virus Infection” from Massachusetts General Hospital (MGH) and reported in the Journal of the American Society of Nephrology.
DATA SHOWS TB INCIDENCE IN POST-RENAL TRANSPLANT POPULATION
Sept. 2 - The incidence of active tuberculosis (TB) in patients following kidney transplants is significantly higher than the general population. While researchers in the past have described this as presenting in the first year following solid organ transplantation and is predominantly caused by disease reactivation.
PANCREATIC ISLET TRANSPLANTATION MAY BE EXTENDED BY MICROGEL DELIVERY METHOD
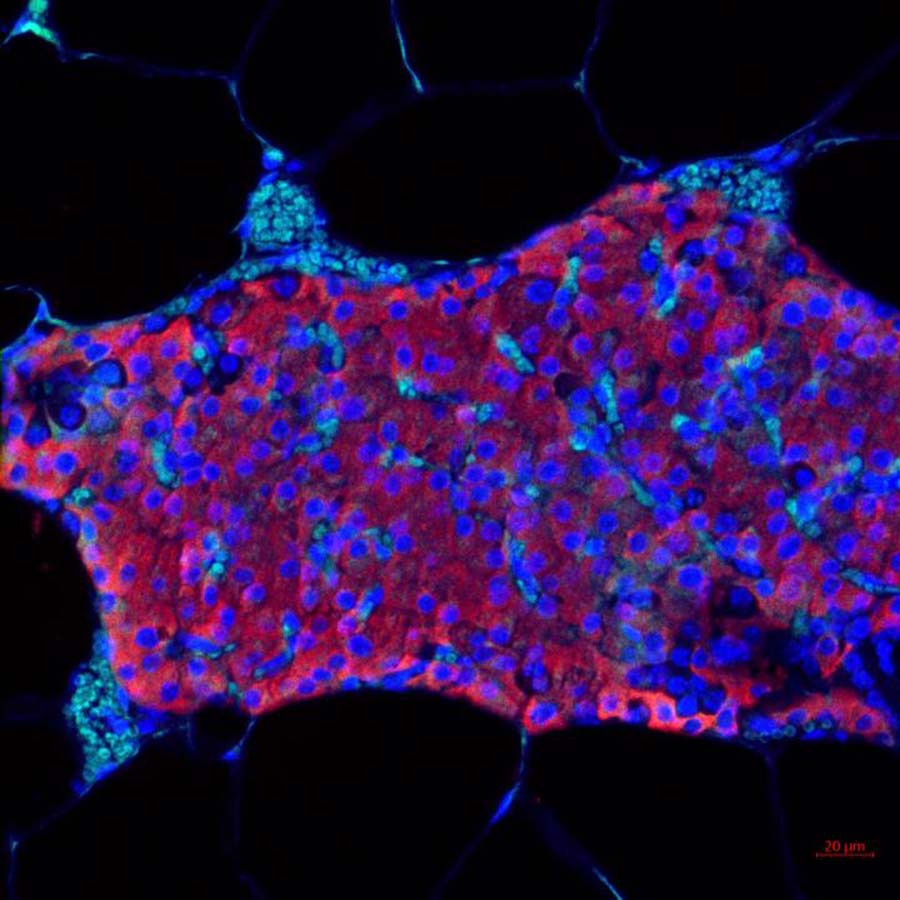
Sept. 1 - In type 1 diabetes, the beta cells of the pancreas no longer make insulin, so a person who has type 1 diabetes must take insulin daily to live. Transplanted islet cells, however, can take over the work of the destroyed cells. Islets are cells found in clusters throughout the pancreas and are made up of several types of cells. One of these is beta cells, which make insulin.
UPCOMING MEETINGS AND ANNOUNCEMENTS
TTS BUSINESS MEETING

In accordance with the TTS By-Laws, a Business Meeting will be held shortly after the close of the TTS 2020 Virtual Congress. The meeting will be help on Sunday, September 20, at 8 AM Montreal time and will last approximately one hour. The agenda will include an update on the TTS finances, the announcement of the TTS 2024 Congress location, a presentation for the TTS 2022 Congress in Buenos Aires, the announcement of the 2020-2022 Councilors and Officers and the official change of TTS Presidency. All members are invited to participate.
As this meeting will be virtually, we will send all members a link to join this meeting shortly before the meeting takes place.
Sincerely,
John Fung
Secretary, The Transplantation Society
18th Asian Pacific Congress of Nephrology (APCN)
Asian Transplantation Week (ATW) 2020
SPLIT Virtual Annual Meeting
FOCIS 2020 Virtual Annual Meeting
TTS members are invited to attend the FOCIS 2020 Virtual Annual Meeting – THE meeting in translational immunology!
The Federation of Clinical Immunology Societies exists to improve human health through immunology.
The FOCIS 2020 Virtual Annual Meeting on October 28-31, 2020, brings together an interdisciplinary group of world-renowned physicians and researchers to share the latest findings on diseases impacting the immune system.
IPITA-IXA-CTRMS Joint Congress
Contact
Address
The Transplantation Society
International Headquarters
740 Notre-Dame Ouest
Suite 1245
Montréal, QC, H3C 3X6
Canada
Используйте Вавада казино для игры с бонусом — активируйте промокод и начните выигрывать уже сегодня!

















